The Lac Operon: A Comprehensive Study to Its Structure and Mechanism
Introduction: A Foundational Discovery in Biology
How does a bacterium make an energy-efficient decision about which sugar to consume? E. coli uses a finely tuned genetic switch called the lac operon, a cluster of genes that controls lactose metabolism. Its discovery in 1961 by François Jacob and Jacques Monod, awarded the Nobel Prize, revealed the fundamental principles of gene regulation (Jacob & Monod, 1961).
The lac operon is one of the most extensively studied gene regulatory systems and serves as a foundational model for understanding transcriptional control in prokaryotes. First described by Jacob and Monod in 1961, it introduced the concept of regulated gene expression and laid the groundwork for modern molecular genetics. Despite its apparent simplicity, the lac operon integrates multiple environmental signals through a dual-control mechanism, making it a powerful example of biological decision-making at the molecular level.
This comprehensive study dissects the elegant structure and precise mechanism of the lac operon. You will see how a simple genetic switch embodies the sophisticated logic of life itself.
The Genetic Blueprint: Architecture of the Lac Operon
First, what is an operon? In bacteria, genes with related functions are grouped under a shared regulatory unit called an operon (Osbourn & Field, 2009). The lac operon is the archetypal example, producing a single polycistronic mRNA that encodes all enzymes necessary for lactose metabolism. It contains all the instructions for lactose metabolism, controlled by a single promoter. This polycistronic organization allows for a rapid, coordinated response to environmental change. The lac operon consists of two main parts: the regulatory region and the structural gene region.
Control Panel: The Regulatory DNA of the Lac Operon
The Control Panel (The Regulatory Region): This region doesn’t code for proteins. Instead, it functions as an integrated circuit of switches and docks.
- The Promoter (lacP): The DNA sequence where RNA polymerase binds to initiate transcription. In simple words, this is the landing pad where RNA polymerase binds to initiate transcription.
- The Operator (lacO): Binding site for the lac repressor. When the repressor is bound, RNA polymerase is physically blocked (Gatti-Lafranconi et al., 2013).
- The CAP Binding Site: When glucose is scarce, cAMP binds CAP, which then binds this site upstream of the promoter, enhancing RNA polymerase recruitment and transcription.
Crucially, a low level of “basal” or “leaky” transcription always occurs, even in the repressed state. This minimal activity is essential—it allows a few molecules of β-galactosidase to be present, ready to convert the first incoming lactose molecules into the inducer, allolactose.
Upstream of the main lac operon lies the separate, constitutively expressed lacI gene. It constantly produces the lac repressor protein, ensuring the system’s default state is firmly “off.”
Note: Click for detail about promoter of a gene, “Promoter of a Gene: Basic Structure and Function in Eukaryotic Cells“
Action Team: The Structural Genes of the Lac Operon
When the control panel is activated, a single polycistronic mRNA is produced, carrying the code for three proteins that function as a perfect metabolic team (see Figure 1). These interacting regulatory sequences and structural genes together constitute a highly coordinated metabolic unit, summarized in Table 1, which details each component of the lac operon and its specific contribution to system function.
| Table 1: The Functional Components of the Lac Operon | |||
| Component | Type | Primary Function | Key Insight |
| lacI Gene | Regulatory | Produces the lac repressor protein. | Sets the default “OFF” state. |
| Promoter (lacP) | Regulatory Sequence | Binding site for RNA polymerase. | The transcription start site. |
| Operator (lacO) | Regulatory Sequence | Binding site for the lac repressor. | The main negative control switch. |
| CAP Site | Regulatory Sequence | Binding site for the CAP-cAMP complex. | The main positive control switch. |
| lacZ Gene | Structural | Encodes β-galactosidase. Cleaves lactose and produces the inducer allolactose. | Performs the core reaction and creates the “on” signal. |
| lacY Gene | Structural | Encodes lactose permease. Actively transports lactose into the cell. | Ensures substrate is available. |
| lacA Gene | Structural | Encodes galactoside transacetylase. Modifies non-metabolizable galactosides. | Non-essential for lactose catabolism; likely a detoxification enzyme. |
| Table 1 provides a concise breakdown of the core genetic elements that constitute the functional lac operon system, highlighting their roles in its integrated logic. | |||
Figure 1 illustrates the structural organization of the lac operon, including its regulatory elements, the polycistronic mRNA transcribed from the operon, and the corresponding protein products that collectively mediate lactose metabolism.

The Dual-Control Mechanism: How the Lac Operon Functions as a Logic Gate
The true genius of the lac operon lies in its two-tiered regulatory mechanism. It integrates two independent environmental signals—lactose and glucose—through separate proteins, creating a sophisticated biological decision-making module.
Negative Control: The Lactose-Sensitive Repressor
This layer is an inducible switch responding to lactose.
- Default OFF State: With no lactose present, the lac repressor binds tightly to the operator, forming a physical blockade. The lac operon is silenced.
- Induction by Allolactose: When lactose enters, existing basal β-galactosidase converts a trace amount into allolactose. This inducer binds the repressor, causing a conformational change that reduces its DNA affinity. The repressor dissociates, removing the roadblock.
Important Note:
Lactose itself is not the direct inducer of the lac operon. A small amount of β-galactosidase converts lactose into allolactose, which binds the lac repressor and induces transcription.
Positive Control: Activation by Glucose Scarcity
The second layer responds to energy status via catabolite repression.
- Glucose High: E. coli‘s preferred fuel is abundant. Intracellular levels of the signaling molecule cyclic AMP (cAMP) are low.
- Glucose Low: cAMP levels surge. cAMP binds to the Catabolite Activator Protein (CAP). The resulting CAP-cAMP complex binds to its target site upstream of the lac operon promoter (Fic et al., 2009). This binding bends the DNA and recruits RNA polymerase, boosting transcription by up to 50-fold.
Key Insight: The lac operon functions as a biological AND gate, integrating two signals: lactose presence (to inactivate the repressor) and low glucose (to activate CAP–cAMP). Importantly, basal transcription occurs even with glucose present, allowing a graded and flexible response to mixed sugar conditions.
Integrated Decision Matrix: The Four States of the Lac Operon
The final transcriptional output is determined by the combined status of both regulatory proteins (See figure 2). This logic is elegantly captured in a simple state table. This integrated control is best understood by examining how different combinations of glucose and lactose availability influence the activity of the repressor and the CAP–cAMP complex, as summarized in Table 2.
| Table 2: The Integrated Logic of Lac Operon Regulation | ||||
| Condition (Glucose / Lactose) | Repressor State | CAP-cAMP State | Operon Activity | Biological Rationale |
| Present / Absent | Bound | Not Bound | OFF | No substrate available. Glucose suffices. |
| Present / Present | Unbound | Not Bound | LOW (Basal) | Lactose is present, but glucose is the preferred fuel. Minimal expression due to catabolite repression. |
| Absent / Absent | Bound | Bound | OFF | Cell is primed for alternative sugars (CAP active), but without lactose, the repressor blocks transcription. |
| Absent / Present | Unbound | Bound | FULLY ON | Ideal conditions: substrate requires digestion, and no better energy source is available. |
| Table 2 summarizes the four possible regulatory outcomes of the lac operon, demonstrating how it functions as an integrated environmental sensor and decision-maker. | ||||
Figure 2 illustrates the functional states of the lac operon under two different conditions: when glucose is present, transcription is repressed despite lactose availability; when glucose is absent, the CAP–cAMP complex activates transcription, enabling full expression of the operon.

Enhanced Mechanistic Insights & Clarifications
While the lac operon is often introduced as a simple on/off switch, its molecular regulation is precise and sophisticated:
- The Role of Allolactose as the True Inducer: Lactose itself is not the direct inducer. Basal β-galactosidase activity converts lactose to allolactose, which binds the lac repressor, causing it to release from the operator. This allows RNA polymerase to transcribe the operon only when lactose is truly available (Jacob & Monod, 1961).
- Polycistronic mRNA: Coordinated Gene Expression: A single mRNA encodes LacZ, LacY, and LacA, ensuring coordinated production of all lactose-metabolizing proteins.
- LacZ (β-galactosidase): Cleaves lactose and produces allolactose.
- LacY (lactose permease): Transports lactose into the cell efficiently.
- LacA (transacetylase): Detoxifies non-metabolizable galactosides; non-essential for lactose catabolism.
- CAP–cAMP: Positive Regulation and Glucose Sensitivity: Low glucose increases cAMP, which binds CAP. The CAP–cAMP complex then enhances RNA polymerase binding and transcription, enabling the cell to prioritize energy-efficient metabolism.
- Integrated AND Gate Logic: Full operon expression requires both lactose presence (repressor inactivated) and low glucose (CAP–cAMP activated). Basal transcription can still occur under intermediate conditions, demonstrating graded, dynamic regulation (Velazco et al., 2021).
- Systems-Level Behavior and Stochasticity: Some populations of E. coli show bistability in operon expression, with cells randomly switching between ON and OFF states. This allows survival flexibility in fluctuating environments, a principle widely used in systems and synthetic biology.
Beyond the Bacterium: The Lac Operon’s Enduring Legacy
The influence of the lac operon stretches from foundational theory to cutting-edge biotechnology and synthetic biology.
- Foundational Concept: It provided the first concrete models for mRNA, repressor proteins, and inducible gene regulation, shaping modern molecular biology (Alberts et al., 2022).
- Indispensable Biotech Tool: The promoter and regulatory elements of the lac operon are engineered into countless expression vectors. Synthetic inducers like IPTG are used globally for precise, high-yield production of proteins, from research enzymes to therapeutic insulin and vaccines (Tan et al., 2012).
- Synthetic Biology Module: Components of the lac operon serve as standard, reliable parts for constructing genetic circuits, biosensors, and inducible CRISPR-Cas systems, enabling the programming of cellular behavior.
Systems Perspective: Modern studies show the lac operon can exhibit bistability and stochastic switching at intermediate inducer concentrations, making it a classic model in systems biology for studying nonlinear dynamics in gene networks (Velazco et al., 2021).
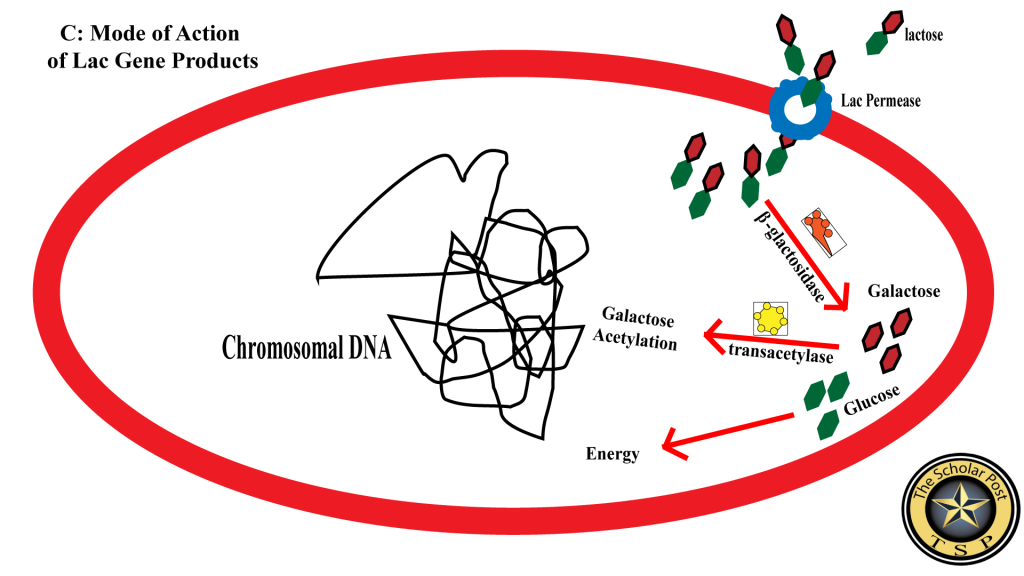
Figure 3 illustrates the mode of action of the lac operon’s structural genes, showing how the products of lacZ, lacY, and lacA work together to metabolize lactose efficiently within the cell.
At intermediate inducer concentrations, lac operon expression can exhibit bistability, resulting in distinct subpopulations of cells with the operon either fully ON or OFF. This stochastic behavior highlights how gene regulation can generate phenotypic diversity even in genetically identical populations.
Conclusion
The lac operon exemplifies evolutionary elegance and regulatory precision. Its compact genetic design and dual-control mechanism ensure that metabolic enzymes are produced efficiently, only when needed. From shaping molecular biology theory to enabling biotechnology applications, the lac operon continues to inspire research and demonstrate nature’s remarkable engineering.
Frequently Asked Questions About the Lac Operon
What is the Lac Operon in E. coli?
The Lac Operon is a gene regulatory system in E. coli that controls lactose metabolism. It includes regulatory and structural genes that respond to lactose and glucose availability, thereby ensuring efficient transcription only when lactose serves as an energy source.
How does the Lac Operon work in the presence of lactose?
When lactose is present, it forms allolactose and inactivates the repressor. Consequently, the Lac Operon becomes active, RNA polymerase binds the promoter, and transcription of lactose-utilizing genes begins.
How is the Lac Operon turned OFF?
In the absence of lactose, the repressor binds the operator region. Therefore, the Lac Operon remains inactive, and RNA polymerase cannot transcribe the structural genes, preventing unnecessary enzyme production.
What is the role of glucose in Lac Operon regulation?
Glucose levels strongly influence the Lac Operon. When glucose is low, cAMP binds CAP and enhances RNA polymerase binding. As a result, transcription efficiency increases in the presence of lactose.
What are the main components of the Lac Operon?
The Lac Operon consists of a promoter, operator, CAP binding site, and three structural genes: lacZ, lacY, and lacA. Together, these elements regulate lactose metabolism in bacteria.
Why is the Lac Operon an inducible operon?
The Lac Operon is inducible because it activates only when lactose is available. Thus, it allows bacteria to conserve energy by producing enzymes only when required.
Why is the Lac Operon important in molecular biology?
The Lac Operon serves as a classic model for understanding gene regulation. Moreover, it explains how cells integrate environmental signals to control transcription efficiently.
References and Further Reading
- Osbourn, A.E., Field, B. Operons. Cell. Mol. Life Sci. 66, 3755–3775 (2009). https://doi.org/10.1007/s00018-009-0114-3
- Gatti-Lafranconi, W. P. Dijkman, S. R. A. Devenish, and F. Hollfelder, “A single mutation in the core domain of the lac repressor reduces leakiness,” Microb. Cell Fact., 2013.
- Fic, E et al., “CAMP receptor protein from escherichia coli as a model of signal transduction in proteins – A review,” Journal of Molecular Microbiology and Biotechnology. 2009.
- Tan, J.S., Ramanan, R.N., Ling, T.C. et al. The role of lac operon and lac repressor in the induction using lactose for the expression of periplasmic human interferon-α2b by Escherichia coli . Ann Microbiol 62, 1427–1435 (2012). https://doi.org/10.1007/s13213-011-0394-3
- S. Velazco et al., “Modeling Gene Expression: Lac operon,” 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Mexico, 2021, pp. 1086-1091, doi: 10.1109/EMBC46164.2021.9630940.
- Jacob, F., & Monod, J. (1961). Genetic regulatory mechanisms in the synthesis of proteins. Journal of Molecular Biology, 3(3), 318–356. https://doi.org/10.1016/S0022-2836(61)80072-7 (Subscription required)
- Alberts, B., Johnson, A., Lewis, J., Morgan, D., Raff, M., Roberts, K., & Walter, P. (2015). Molecular biology of the cell(6th ed.). Garland Science.
- For the latest edition: Alberts, B., Heald, R., Johnson, A., Morgan, D., Raff, M., Roberts, K., & Walter, P. (2022). Molecular biology of the cell (7th ed.). W. W. Norton & Company.