Childhood Brain Tumors Reprogram Skull Bone Marrow to Escape Immunity
Quick Takeaways
Families. A future with kinder treatments is coming into view. Instead of bombarding the entire body, researchers are now targeting the skull itself—where the tumor secretly builds its defenses. This approach is still years from the clinic, but for families searching for hope, it’s a profound step in the right direction.
Science Readers. This is the first evidence that skull marrow educates immune cells—and in pediatric brain tumors, the lesson is betrayal. Cerebrospinal fluid carries signals to the skull, where they reprogram immune cells into tumor protectors. It’s immunology rewritten, with real therapeutic stakes.
Clinicians. The implications are immediate and actionable. Cytokines in the CSF could become a powerful biomarker. Blocking GM-CSF reverses immune suppression in mouse models. And combined with checkpoint inhibitors? That synergy could finally unlock responses where single agents have failed.
Share This Discovery
“Childhood brain tumors don’t just evade the immune system—they actively train it to tolerate them.”
Introduction
Every year, more than 4,000 children in the United States alone are diagnosed with brain tumors, making them the most common solid tumors of childhood and the leading cause of childhood cancer death. Yet effective treatments have remained frustratingly elusive. Traditional immunotherapies—treatments that harness the body’s immune system to fight cancer—have shown limited success in pediatric brain tumors, leaving researchers searching for answers about why these aggressive cancers evade immune detection.
Now, a groundbreaking study published in Nature Genetics in 2026 reveals an unexpected culprit: the cranial bone marrow itself. Cooper et al. (2026) discovered that childhood brain tumors actually “teach” nearby skull bone marrow to create a protective immune environment, essentially convincing the body to tolerate the cancer rather than attack it.
This remarkable finding opens entirely new possibilities for treatment—though it’s important to note that these discoveries currently remain in preclinical mouse models, and human translation will require further study.
Understanding Childhood Brain Tumors: A Primer
Before diving into the discovery, it helps to understand what childhood brain tumors are and why they’re so challenging. Brain tumors arise from different cell types within the central nervous system and are classified based on their cell of origin, location, and molecular characteristics (Louis et al., 2021). They are the most common solid tumors in children, accounting for 20-25% of all childhood cancers, second only to leukemia in overall cancer incidence (Ostrom et al., 2022).
The table below summarizes the most common types of childhood brain tumors, their characteristics, and outcomes. This diversity is important because different tumors may respond differently to immune therapy. The Cooper study suggests the skull marrow mechanism may apply across multiple types, including ependymoma, medulloblastoma, and choroid plexus carcinoma.
| Table 1: Common Types of Childhood Brain Tumors | |||||
| Tumor Type | Prevalence | Typical Age | Location | 5-Year Survival | Key Molecular Markers |
| Medulloblastoma | 18-25% | <10 years | Cerebellum | 63-95% by subtype | WNT, SHH, Group 3, Group 4 |
| Pilocytic Astrocytoma | 15-20% | First 2 decades | Cerebellum | >95% | BRAF alterations |
| Ependymoma | 5-10% | Mean 4-6 years | Posterior fossa, supratentorial | 50-85% | ZFTA-RELA fusion (ST), PFA (PF) |
| ZFTA-RELA Ependymoma (study focus) | Majority of ST ependymomas | Children/young adults | Supratentorial | ~50-60% | ZFTA-RELA fusion gene |
| Diffuse Intrinsic Pontine Glioma (DIPG) | 10-15% | 5-10 years | Pons | <10% | H3K27M mutation |
| Choroid Plexus Tumors | 2-4% | <2 years | Lateral ventricle | 30-100% | TP53 mutations (carcinoma) |
| Germ Cell Tumors | 3-5% | Adolescents | Pineal, suprasellar | 70-90% | KIT/RAS mutations |
| Atypical Teratoid/Rhabdoid Tumor (AT/RT) | 1-2% | <3 years | Posterior fossa | 20-50% | SMARCB1/INI1 loss |
| Sources: Ostrom et al. (2022); Louis et al. (2021); Northcott et al. (2019); Pajtler et al. (2015); Mackay et al. (2017) | |||||
As the table illustrates, childhood brain tumors are remarkably diverse, with outcomes ranging from excellent (pilocytic astrocytoma, >95% survival) to devastating (DIPG, <10% survival). This diversity reflects the different cells of origin from primitive embryonal cells in medulloblastoma to glial cells in gliomas and ependymal cells in ependymomas (Northcott et al., 2019).
Glossary of Key Terms
| Term | Simple Explanation |
| CSF (Cerebrospinal Fluid) | Clear fluid that bathes the brain and spinal cord |
| HSPC (Hematopoietic Stem and Progenitor Cell) | “Master cells” in bone marrow that produce blood and immune cells |
| GM-CSF | A signaling protein that stimulates production of immune cells |
| Treg (Regulatory T cell) | Immune cell that suppresses immune responses |
| MHC-II | Molecular “plate” that presents antigens to T cells |
| MDSC (Myeloid-Derived Suppressor Cell) | Immune cell that blocks cancer-killing immune responses |
What Was the Problem?
The brain has traditionally been viewed as “immune-privileged”—meaning it exists somewhat separate from the body’s normal immune surveillance. However, recent research has challenged this concept. Cooper et al. (2026) recognized that if we could understand how childhood brain tumors manipulate the immune system, we might unlock better treatment approaches.
The research gap was significant: while scientists knew that cerebrospinal fluid (CSF) flows directly to cranial bone marrow through specialized channels, carrying brain-derived signals (Mazzitelli et al., 2022), no one had fully understood how these signals influence blood cell production (hematopoiesis) in the context of cancer.
The key question was whether childhood brain tumors could be sending messages through the CSF that reprogram local immune development.
To understand the broader context of immune therapy in cancer, it helps to recognize that most immunotherapies target the tumor itself. This study takes a completely different approach by targeting the source of immune cells.
How Was the Study Conducted?
The following steps were applied in this study by Cooper et al. (2026).
Building a Mouse Model of Childhood Brain Tumors
The research team first created a genetically engineered mouse model of ZFTA-RELA ependymoma. This is a specific type of childhood brain tumor that accounts for the majority of supratentorial ependymomas in children (Pajtler et al., 2015). They achieved this by inserting the fusion gene responsible for this cancer into embryonic day 9.5 radial glia cells, the cells that originally give rise to these tumors. This approach reliably produces tumors with molecular features identical to those seen in children.
Tracking Immune Cells with Flow Cytometry
The team used flow cytometry to identify and count millions of individual immune cells based on specific surface markers. They collected samples from tumor tissue, cranial bone marrow, peripheral bone marrow from leg bones, dura mater (the brain’s protective outer layer), and cerebrospinal fluid. A clever technical twist made this possible. The team injected a CD45 antibody into the bloodstream just before collecting the samples. This allowed them to distinguish between cells actually residing in the tissues and those merely passing through.
Single-Cell RNA Sequencing Analysis
Using single-cell RNA sequencing, the team then mapped the genetic activity of over 125,000 individual cells from tumor-bearing and control mice. They also analyzed human fetal brain tissue and 14 different types of childhood brain tumors, confirming their mouse findings translated to human disease.
Testing CSF Signaling
To determine whether brain-derived signals reach cranial bone marrow, Cooper et al. (2026) injected fluorescent antibodies into the cerebrospinal fluid. Within just two hours, these antibodies had labeled 85% of hematopoietic stem and progenitor cells (HSPCs) in the cranial bone marrow, along with 60% of those in the dura mater—confirming that skull marrow sits directly in the path of brain-derived signals.
Transfer Experiments
Finally, in elegant transfer experiments, CSF from tumor-bearing mice was injected into healthy mice. Within six hours, these healthy recipients showed the same immune changes seen in tumor-bearing animals. Their skull marrow and brain now contained increased numbers of neutrophils, monocytes, HSPCs, and B cells. This demonstrated that something in the tumor-conditioned CSF, not the tumor itself, was reprogramming local hematopoiesis.
Why This Discovery Is Unusual
The discovery is unusual because bone marrow is typically studied as a distant immune organ, located far from the sites it serves. This study instead reveals that the skull bone marrow acts as a local immune interface with the brain, directly receiving signals from cerebrospinal fluid through specialized channels.
Such direct communication between the brain and bone marrow was only discovered recently (Mazzitelli et al., 2022). What makes the Cooper study groundbreaking is demonstrating that childhood brain tumors actively exploit this connection to create a protective immune environment. The tumor isn’t passively evading detection. It’s actively reprogramming the skull’s immune factory, training it to produce cells that offer protection. This represents a fundamental shift in how we understand brain tumor immunity and opens an entirely new avenue for therapeutic intervention.
Figure 1. Tumor–Skull Marrow Communication
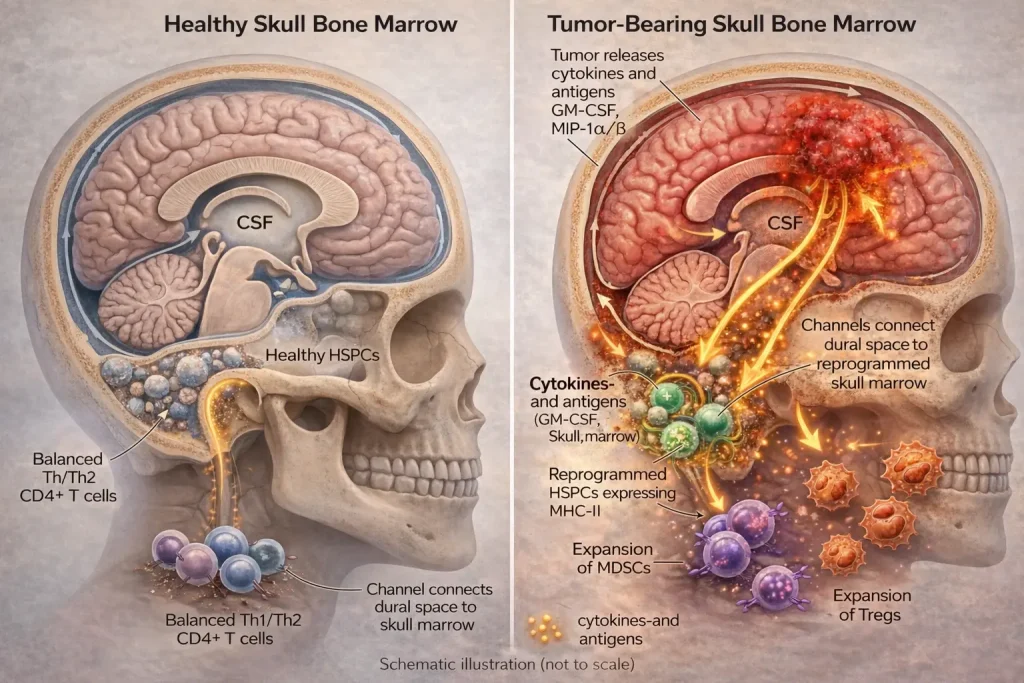
Brain tumors release cytokines and antigens into cerebrospinal fluid (CSF), which travel through skull–meningeal channels to the skull bone marrow. These signals activate hematopoietic stem and progenitor cells (HSPCs), driving expansion of immunosuppressive cells such as regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs), enabling tumor immune evasion. The left panel shows the healthy state with balanced immune cells.
What Did Researchers Discover?
The following key discoveries emerged from the study by Cooper et al. (2026).
Blood Stem Cells Become Antigen Presenters
The researchers found that hematopoietic stem and progenitor cells within brain tumors expressed Major Histocompatibility Complex class II (MHC-II) molecules. These molecules are the cellular machinery typically used by professional antigen-presenting cells to show foreign material to T cells. Remarkably, over 90% of HSPCs in ZFTA-RELA tumors expressed MHC-II at levels comparable to dendritic cells, the body’s professional antigen presenters. This matters for childhood brain tumors because primitive blood stem cells are being recruited to participate in immune responses, fundamentally altering how the immune system perceives the cancer.
Skull Marrow Responds Differently Than Leg Marrow
Using EdU labeling, which marks dividing cells, the researchers showed dramatically increased proliferation of HSPCs in cranial marrow of tumor-bearing mice, while tibial (leg) marrow remained unchanged. This skull-specific response extended to multiple childhood brain tumor types i.e., ependymoma, choroid plexus carcinoma, and group 3 medulloblastoma. This matters for childhood brain tumors because it appears to be a general phenomenon across different tumor types, not limited to one specific cancer, suggesting broad therapeutic potential.
CSF Carries Specific Signals
Proteomic analysis of CSF revealed significantly elevated levels of multiple cytokines in tumor-bearing mice. These included GM-CSF (granulocyte-macrophage colony-stimulating factor), which increased five-fold; G-CSF, which increased three-fold; MIP-1a/b chemotactic signals; and IL-12/23 and IL-4, which are T cell polarization signals. When the team blocked cell migration using AMD3100, they trapped immune cells in skull marrow, preventing their movement to the tumor. This matters for childhood brain tumors because tumor-derived CSF signals both instruct skull marrow hematopoiesis and direct cell migration to the tumor site, creating a complete communication loop. Understanding cerebrospinal fluid biology is essential to appreciating how these signals travel.
Skull HSPCs Drive Treg Polarization
Skull marrow HSPCs from tumor-bearing mice actively presented antigens to CD4+ T cells, but instead of promoting an inflammatory response, they drove polarization toward regulatory T cells (Tregs)—immune cells that suppress rather than activate immune responses. When they tested a tumor-specific neoantigen, a peptide unique to the ZFTA-RELA fusion protein, they observed the same Treg polarization in tumor-bearing mice—the childhood brain tumor antigen was treated as “self.” In healthy mice without tumors, the same antigen triggered a normal inflammatory Th1 response instead. This matters for childhood brain tumors because it represents a powerful immunosuppressive mechanism specific to the tumor environment—the tumor actively teaches the immune system to tolerate it.
Bone Marrow Is Fundamentally Reprogrammed
Using single-nuclear ATAC sequencing, which measures which genes are accessible for activation, the team showed that skull HSPCs from tumor-bearing mice underwent fundamental reprogramming. Their chromatin landscapes shifted toward producing myeloid cells at the expense of lymphoid development, biasing the immune system toward suppressive rather than attacking cells. HSPCs produced significantly more granulocyte-macrophage colonies and differentiated toward myeloid-derived suppressor cell (MDSC)-like phenotypes characterized by IL-10, nitrite, and reactive oxygen species production. This matters for childhood brain tumors because the tumor doesn’t just temporarily influence immune cells. It permanently reprograms the bone marrow’s “factory settings” toward suppression, creating a sustained effect.
Blocking GM-CSF Causes Tumor Regression in Mice
Building on these insights, Cooper et al. (2026) tested whether disrupting this skull marrow-tumor axis could have therapeutic benefit. They treated tumor-bearing mice with a single injection of antibodies against GM-CSF or its receptor (GM-CSFRA/mavrilimumab) directly into the CSF.
The results in these preclinical models were remarkable. Profound tumor regression was sustained for approximately six weeks, with over threefold increase in survival time. Treatment decreased CSF GM-CSF levels, reduced skull HSPC proliferation, diminished tumor-associated suppressive myeloid cells, and increased cancer-killing CD8+ T cell infiltration.
When combined with anti-CTLA-4 immune checkpoint blockade, efficacy improved further. Intravenous administration also proved effective, which could simplify potential clinical translation.
| Clinical Trial Readiness Scale | ||
| Phase | Status | Timeline Estimate |
| Preclinical mouse studies | Complete | 2026 |
| Safety and toxicology studies | Not started | 1-2 years |
| Phase I human trials | Not started | 2-4 years |
| Phase II efficacy trials | Not started | 4-6 years |
| Phase III pivotal trials | Not started | 6-8 years |
| Clinical availability | Not started | 8-10 years |
Study Limitations
Although these findings are exciting, several important limitations must be acknowledged. The experiments were conducted in mouse models, and immune responses in humans can differ significantly from those in mice. The precise mechanisms by which human skull bone marrow responds to brain tumors may vary, and the specific antigens driving immune tolerance in pediatric patients remain to be identified.
In addition, long-term effects of GM-CSF blockade on normal immune development in children remain completely unknown. Because the immune system continues to mature throughout childhood, interventions that modulate hematopoiesis could have unintended consequences on overall immune competence.
Finally, the study focused on three tumor types—ependymoma, medulloblastoma, and choroid plexus carcinoma. Whether similar mechanisms operate in other pediatric brain tumors such as diffuse intrinsic pontine glioma (DIPG) or high-grade glioma requires further investigation.
Clinical trials will be necessary to determine safety, optimal dosing, and efficacy in pediatric patients before this approach can reach the clinic.
Therapeutic Implications: A New Approach for Childhood Brain Tumors
The Current Treatment Landscape
The current treatment landscape for childhood brain tumors involves several approaches, each with its own benefits and concerns. Surgery removes visible tumor but risks neurological damage. Radiation kills residual cells but can cause severe neurocognitive deficits, especially in children under three years. Chemotherapy provides systemic effect but carries risks of developmental toxicity and secondary cancers.
The proposed GM-CSF blockade offers a fundamentally different approach by reprogramming the immune environment rather than directly attacking the tumor. Because it targets skull marrow rather than the whole body, it could potentially have minimal toxicity compared to current treatments.
This matters for childhood brain tumors because current treatments carry devastating long-term consequences for developing brains. The developing brain is particularly vulnerable to radiation-induced injury, which can cause severe neurocognitive deficits (Merchant et al., 2022). Targeting skull marrow hematopoiesis could represent a fundamentally different, potentially less toxic approach—though this remains to be proven.
How GM-CSF Blockade Works
Figure 2. Therapeutic Mechanism of GM-CSF Blockade (to be uploaded soon)
Why Does It Matter?
This research fundamentally changes how we think about childhood brain tumors and immunity. Rather than viewing the skull as simply protective bone, Cooper et al. (2026) reveal it as an active immunological interface that brain tumors hijack for their own protection.
Previous work has established that CSF can access skull bone marrow through specialized channels (Mazzitelli et al., 2022; Pulous et al., 2022), and that skull marrow serves as a reservoir for myeloid cells responding to brain pathologies (Cugurra et al., 2021; Herisson et al., 2018). This study connects these findings in the context of cancer for the first time.
Real-World Implications
- New therapeutic target. The GM-CSF signaling axis represents an immediately druggable target. Mavrilimumab is already in clinical development for other conditions, potentially accelerating the path to pediatric trials.
- Potential for less toxic treatment. Unlike radiation and chemotherapy that affect the whole body, targeting skull marrow hematopoiesis could be more focused with fewer side effects.
- Immunotherapy combination potential. The synergy with checkpoint blockade suggests that normalizing skull marrow output could make tumors susceptible to existing immunotherapies that have shown limited efficacy in pediatric brain tumors (Majzner et al., 2022).
- Diagnostic implications. CSF cytokine profiling might eventually help identify which patients could benefit from this strategy, enabling personalized immunotherapy.
What This Means for Different Audiences
For Families
The bottom line is that scientists have discovered that childhood brain tumors hide from the immune system by sending signals to the skull bone marrow. Blocking one of these signals, GM-CSF, made tumors shrink dramatically in mice. This is not a treatment available today—it will take years of testing in humans. However, it represents a genuine new direction for research that offers real hope for kinder, more effective treatments in the future.
Families should continue with proven treatments under their medical team’s guidance and stay informed about clinical trials through resources like the Children’s Oncology Group and CureSearch.
For Researchers
This study opens multiple new avenues for investigation. These include researching CSF cytokines as biomarkers for immune status, exploring GM-CSF blockade in other brain tumor types, combining this approach with existing immunotherapies such as checkpoint inhibitors and CAR-T, and identifying which patients might benefit most.
Key questions to address include the optimal timing and dosing, the durability of response, whether human-equivalent antigens can be identified, and the long-term effects of skull marrow modulation. The skull marrow-HSPC-Treg axis represents an entirely new therapeutic target in neuro-oncology.
For Clinicians
While not yet ready for clinical use, this research suggests several important considerations. CSF cytokine profiling might eventually help identify patients with tumor-induced immune suppression. The GM-CSF pathway is particularly promising for therapeutic intervention. Mavrilimumab, which targets GM-CSFRA, is already in clinical development for other conditions and could potentially be repurposed.
This study should be discussed when counseling families about the future of pediatric neuro-oncology, as it represents a paradigm shift in understanding brain tumor immunity.
What Comes Next?
Cooper et al. (2026) acknowledge several critical next steps, and the broader field points toward additional considerations.
Identifying Specific Antigens
Further work must define precisely which peptides drive immune tolerance in human patients. This could inform vaccine-based or peptide-based immunotherapeutics.
Timing and Durability
Understanding optimal treatment timing and whether repeated dosing could maintain responses requires investigation. It remains unknown whether patients will need one dose or maintenance therapy.
Combination Strategies
Exploring optimal combinations with existing therapies, including other checkpoint inhibitors, will be essential. Current standard treatments for medulloblastoma involve multimodal approaches including surgery, risk-adapted craniospinal irradiation, and chemotherapy (Gajjar et al., 2021). Integrating GM-CSF blockade with these approaches will require careful study.
Clinical Translation
Moving from mouse models to human trials represents the ultimate goal. Fortunately, molecular characterization of childhood brain tumors has advanced dramatically with the 2021 WHO classification (Louis et al., 2021), which now incorporates genetic markers like ZFTA-RELA fusions, histone H3 mutations, and molecular subgrouping of medulloblastoma. This molecular framework will enable precise patient selection for future trials.
Drug Repurposing Potential
Mavrilimumab is already in clinical development for other conditions such as rheumatoid arthritis and giant cell arteritis. This existing safety data could accelerate pediatric trials by 2-3 years compared to developing a new drug from scratch.
Summary: Key Takeaways
- What did this study discover? Childhood brain tumors send signals through CSF to skull bone marrow, reprogramming HSPCs to create immune tolerance.
- How does it work? Tumor-derived GM-CSF drives HSPCs toward myeloid lineage, and HSPCs present antigens and polarize CD4+ T cells to Tregs.
- Is this a cure? No—it is a preclinical discovery in mice. Human trials are needed.
- Why is this exciting? It identifies a completely new therapeutic target, skull marrow hematopoiesis, that could be less toxic than current treatments.
- What is the key therapeutic insight? Blocking GM-CSF signaling disrupted this axis and caused profound tumor regression in multiple mouse models.
- When will this be available? Years away, approximately 8-10 years if successful. However, mavrilimumab is already in clinical development for other conditions.
- Which tumors might respond? Ependymoma, medulloblastoma, and choroid plexus carcinoma in mouse models, and potentially others.
- How does this differ from current immunotherapy? It targets the source of immune cells, the skull marrow, rather than the tumor itself.
Related Resources
Children’s Oncology Group provides information about clinical trials for pediatric cancers. CureSearch for Children’s Cancer offers family resources and research updates. The National Cancer Institute provides patient education on childhood brain tumors. The Pediatric Brain Tumor Foundation offers support and research funding. PubMed can be used to search for updates on Cooper et al. and related studies.
Conclusion
Cooper et al. (2026) have illuminated an entirely new mechanism by which childhood brain tumors establish immune privilege. By demonstrating that tumor-derived CSF cues instruct cranial bone marrow hematopoiesis, driving myeloid bias and Treg polarization, they have identified a therapeutic vulnerability at the very source of local immune cell production.
The finding that a single dose of GM-CSF-blocking antibodies can induce profound tumor regression across multiple aggressive childhood brain tumor models in mice offers genuine hope for families facing these devastating diagnoses. However, it is essential to recognize that these are preclinical findings—the path from mouse models to effective human therapies is long, and many promising approaches fail along the way.
As this research moves toward clinical application with appropriate caution and rigorous testing, it may fundamentally change how we treat the leading cause of childhood cancer death through innovative immune therapy approaches.
The skull, it turns out, is not just protecting the brain—it is actively participating in brain health and disease. And now, we are beginning to understand the conversation.
References
Main Research Article
Cooper, E., Posner, D.A., Lee, C.Y.C., Hu, L., Bonner, S., Taylor, J.T., Baldwin, O., Jimenez-Guerrero, R., Masih, K.E., WickhamRahrmann, K., Eigenbrod, J., Ngo, G., Roamio Franklin, V.N., D’Santos, C.S., Mair, R., Santarius, T., Craven, C., Jalloh, I., Moreno Vicente, J., Halim, T.Y.F., Wang, L., Kreigstien, A.R., Wainwright, B., Swartling, F.J., Khan, J., Clatworthy, M.R., & Gilbertson, R.J. (2026). Childhood brain tumors instruct cranial hematopoiesis and immunotolerance. Nature Genetics, 58(2), 317–328. https://doi.org/10.1038/s41588-025-02499-2
Supporting Articles
- Herisson, F., Frodermann, V., Courties, G. et al. Direct vascular channels connect skull bone marrow and the brain surface enabling myeloid cell migration. Nat Neurosci 21, 1209–1217 (2018). https://doi.org/10.1038/s41593-018-0213-2 (Institutional access may be required; preprint available on PubMed Central)
- Cugurra, A., Mamuladze, T., Rustenhoven, J., Dykstra, T., Beroshvili, G., Greenberg, Z.J., Baker, W., Papadopoulos, Z., Drieu, A., Blackburn, S., Kanamori, M., Brioschi, S., Herz, J., Schuettpelz, L.G., Colonna, M., Smirnov, I., & Kipnis, J. (2021). Skull and vertebral bone marrow are myeloid cell reservoirs for the meninges and CNS parenchyma. Science, 373(6553), eabf7844. https://doi.org/10.1126/science.abf7844 (Institutional access may be required; abstract freely available)
- Mazzitelli, J.A., Smyth, L.C.D., Cross, K.A. et al. Cerebrospinal fluid regulates skull bone marrow niches via direct access through dural channels. Nat Neurosci 25, 555–560 (2022). https://doi.org/10.1038/s41593-022-01029-1(Institutional access may be required)
- Pulous, F.E., Cruz-Hernández, J.C., Yang, C. et al. Cerebrospinal fluid can exit into the skull bone marrow and instruct cranial hematopoiesis in mice with bacterial meningitis. Nat Neurosci 25, 567–576 (2022). https://doi.org/10.1038/s41593-022-01060-2(Institutional access may be required)
Childhood Brain Tumor Epidemiology and Classification
- Louis, D.N., Perry, A., Wesseling, P., Brat, D.J., Cree, I.A., Figarella-Branger, D., Hawkins, C., Ng, H.K., Pfister, S.M., Reifenberger, G., Soffietti, R., von Deimling, A., & Ellison, D.W. (2021). The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro-Oncology, 23(8), 1231-1251. https://doi.org/10.1093/neuonc/noab106 (Open access)
- Ostrom, Q.T., Price, M., Neff, C., Cioffi, G., Waite, K.A., Kruchko, C., & Barnholtz-Sloan, J.S. (2022). CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2015-2019. Neuro-Oncology, 24(Supplement 5), v1-v95. https://doi.org/10.1093/neuonc/noac202 (Open access)
Medulloblastoma
- Northcott, P.A., Robinson, G.W., Kratz, C.P., Mabbott, D.J., Pomeroy, S.L., Clifford, S.C., Rutkowski, S., Ellison, D.W., Malkin, D., Taylor, M.D., Gajjar, A., & Pfister, S.M. (2019). Medulloblastoma. Nature Reviews Disease Primers, 5(1), 11. https://doi.org/10.1038/s41572-019-0063-6 (Institutional access may be required)
- Gajjar, A., Robinson, G. W., Smith, K. S., Lin, T., Merchant, T. E., Chintagumpala, M., Mahajan, A., Su, J., Bouffet, E., Bartels, U., Schechter, T., Hassall, T., Robertson, T., Nicholls, W., Gururangan, S., Schroeder, K., Sullivan, M., Wheeler, G., Hansford, J. R., Kellie, S. J., … Northcott, P. A. (2021). Outcomes by Clinical and Molecular Features in Children With Medulloblastoma Treated With Risk-Adapted Therapy: Results of an International Phase III Trial (SJMB03). Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 39(7), 822–835. https://doi.org/10.1200/JCO.20.01372
Ependymoma
- Larrew, T., Saway, B. F., Lowe, S. R., & Olar, A. (2021). Molecular Classification and Therapeutic Targets in Ependymoma. Cancers, 13(24), 6218. https://doi.org/10.3390/cancers13246218
Pediatric High-Grade Glioma and DIPG
- Mackay, A., Burford, A., Carvalho, D., Izquierdo, E., Fazal-Salom, J., Taylor, K.R., Bjerke, L., Clarke, M., Vinci, M., Nandhabalan, M., Temelso, S., Popov, S., Molinari, V., Raman, P., Waanders, A.J., Han, H.J., Gupta, S., Marshall, L., Zacharoulis, S., … & Jones, C. (2017). Integrated Molecular Meta-Analysis of 1,000 Pediatric High-Grade and Diffuse Intrinsic Pontine Glioma. Cancer Cell, 32(4), 520-537. https://doi.org/10.1016/j.ccell.2017.08.017 (Institutional access may be required; abstract freely available)
- Majzner, R.G., Ramakrishna, S., Yeom, K.W. et al. GD2-CAR T cell therapy for H3K27M-mutated diffuse midline gliomas. Nature 603, 934–941 (2022). https://doi.org/10.1038/s41586-022-04489-4 (Institutional access may be required; abstract freely available)
Treatment and Late Effects
- Merchant, T. E., Pollack, I. F., & Loeffler, J. S. (2010). Brain tumors across the age spectrum: biology, therapy, and late effects. Seminars in radiation oncology, 20(1), 58–66. https://doi.org/10.1016/j.semradonc.2009.09.005






